Remune™ – Your partner in the nutritional management of oncological patients
Product description
Remune™ is a partially balanced Food for Special Medical Purposes (FSMP) for the dietary management of disease-related malnutrition in patients with pre-cachexia or cachexia especially due to cancer or COPD and for patients undergoing chemotherapy.
Remune™ is a supplement to be used in addition to normal food or in combination with standard oral nutrition supplements (ONS).
Remune™ aims to increase the intake of polyunsaturated omega-3 fatty acids from fish oil and vitamin D3 together with energy and protein in order to supply patients with sufficient amounts of nutrients which they may not get through their normal diet. Remune™ is not suitable as the sole source of nutrition.
Why Remune™?
You are looking for a tasty supplement in compliance with the ESPEN and ASPEN guidelines for nutrition in oncology? Remune™ has a smoothie-like texture and is very tasty.
Moreover, it contains EPA and DHA from low oxidized fish oil to supplement the nutritional needs of cancer patients at risk of malnutrition or with cachexia and pre-cachexia.
A closer look at the content of Remune™ and its benefits
Dosage and Age groups: 2 units / day, unless otherwise specified by a clinician or dietician. Suitable for children > 4 years, adults and elderly.
Drink Remune™ directly out of the container with the provided straw or pour into a glass. Available in raspberry and peach flavor.
Special features:
- Partially balanced
- Fruit-juice-based emulsion (86% fruit content)
- High content of EPA & DHA from fish oil (2000 mg / pack)
- High content of vitamin D
- High biological value whey protein
- Contains 9.6 g of protein / pack
- Smoothie-like, fresh and fruity taste
- Flavors: Peach and Raspberry
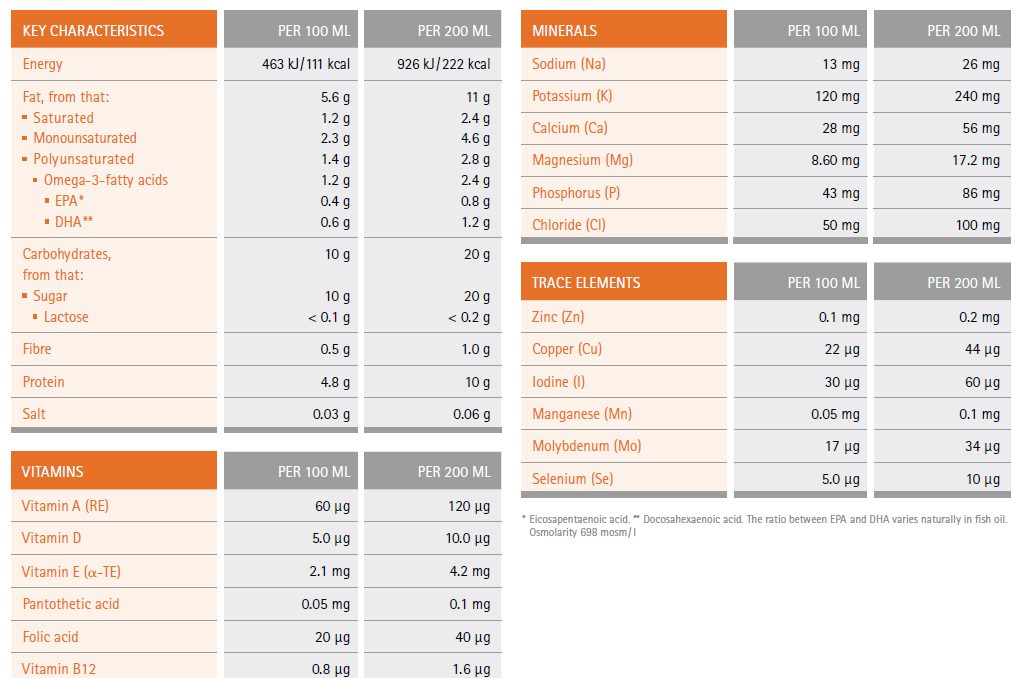
Nutritional details of Remune™
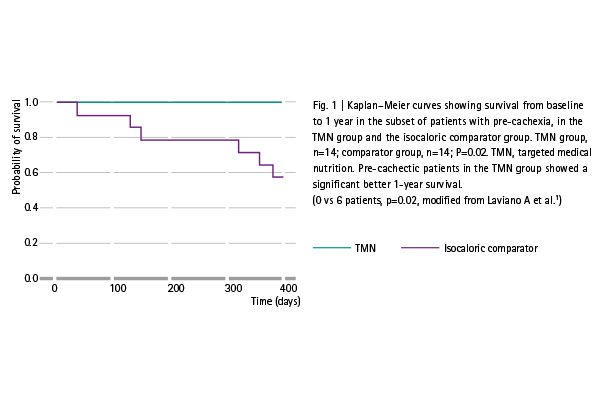
Compared to an isocaloric milk-based drink patients nourished with Remune™ showed:
- Potentially better survival in the subset of patients with pre-cachexia (post-hoc analysis, Fig. 1)
- No negative effect on tolerability of the platinum-based chemotherapy treatment1
- Good tolerability – numerical smaller number of adverse events compared to the comparator group (64 vs. 87 AE)1
- Increased omega-3 fatty acids (EPA and DHA) and vitamin D levels in plasma
Highlights
References
1 Laviano A, Calder PC, Schols AMWJ, Lonnqvist F, Bech M, Muscaritoli M. Safety and Tolerability of Targeted Medical Nutrition for Cachexia in Non-Small-Cell Lung Cancer: A Randomized, Double-Blind, Controlled Pilot Trial. Nutr Cancer. 2020;72(3):439-450.