No content results match your keyword.
Content
You have successfully logged out.
Not registered yet?
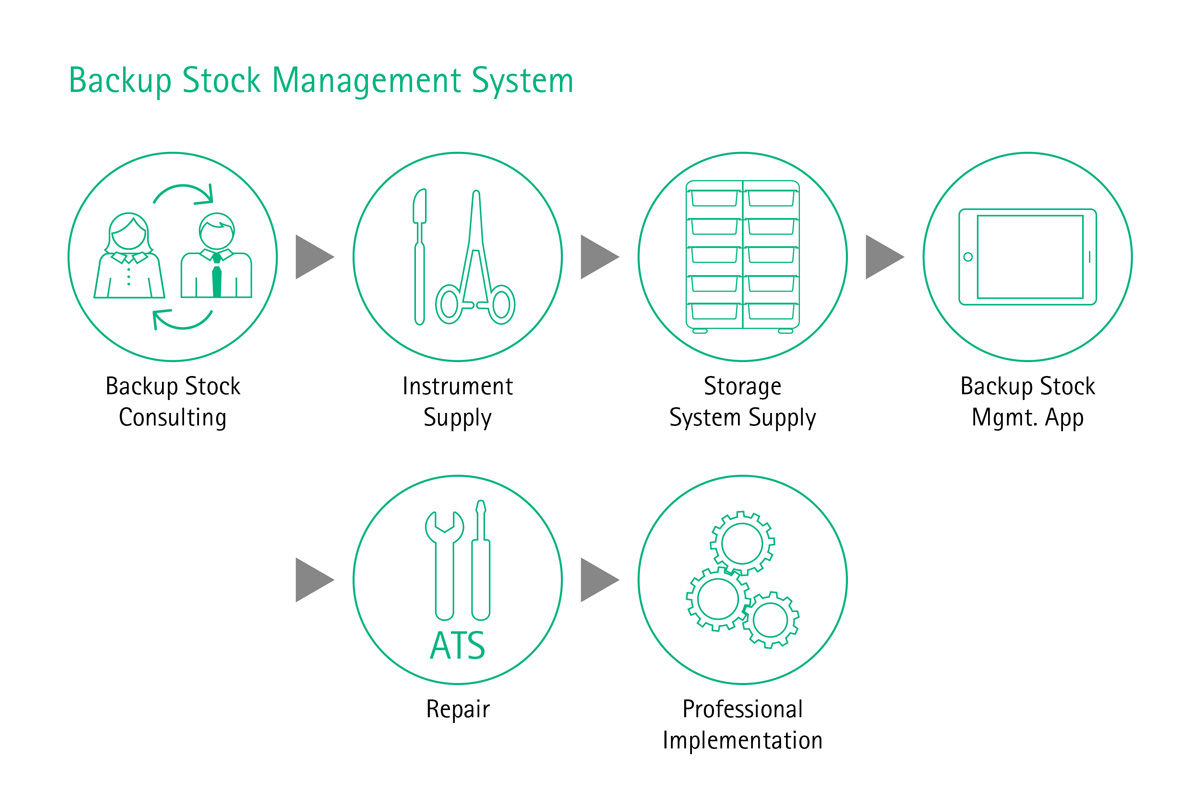
Surgical Asset & Supply Consulting
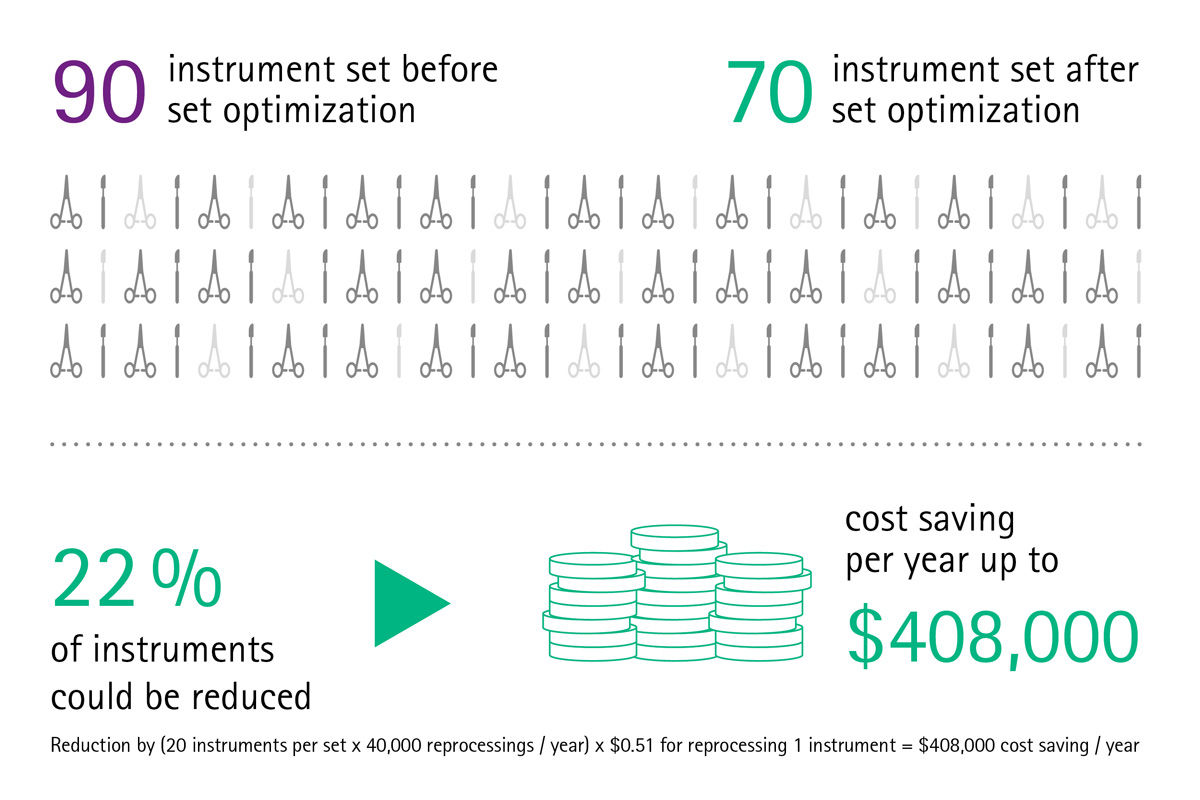
Are you aware of all cost traps in your hospital?


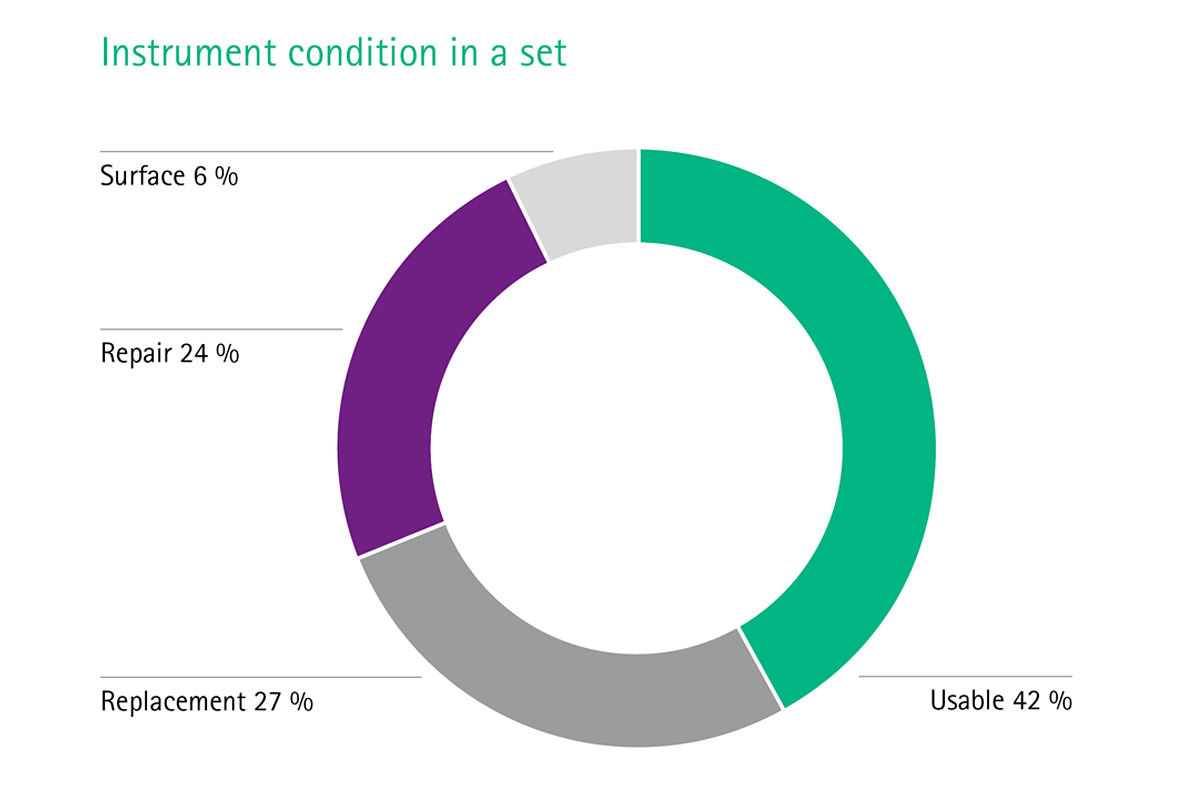
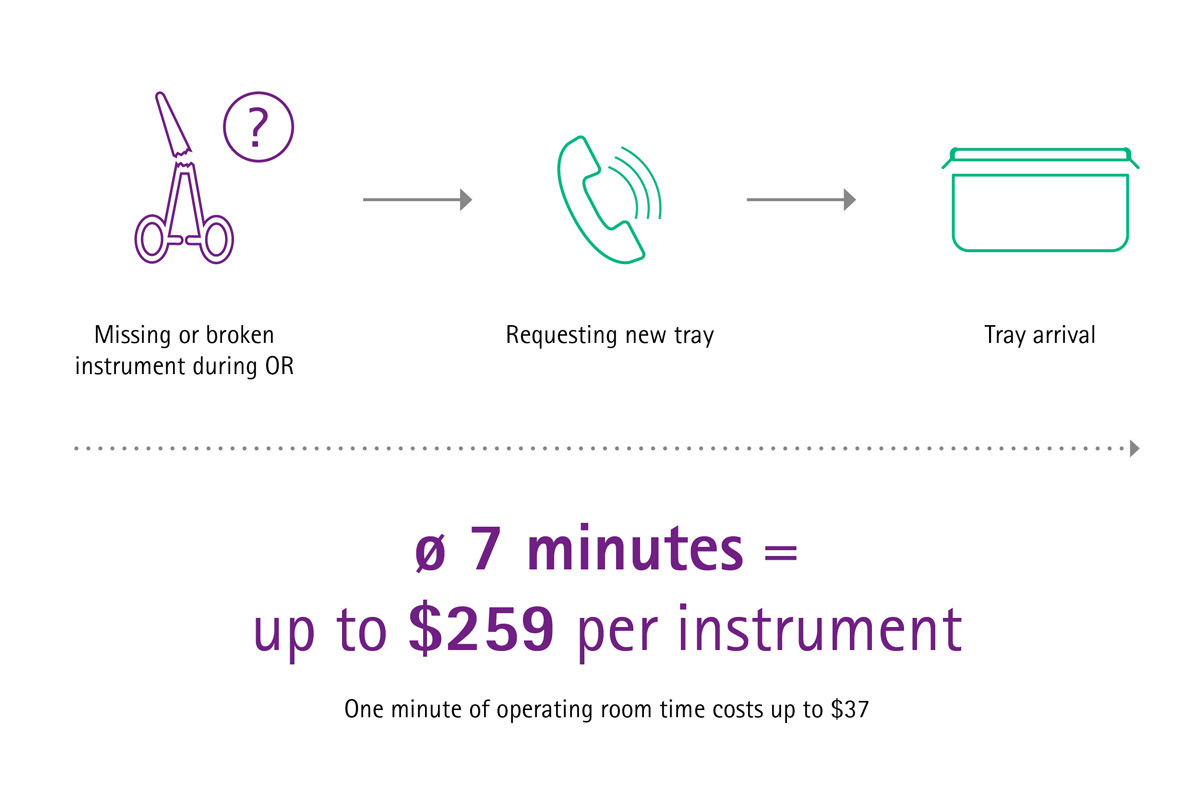
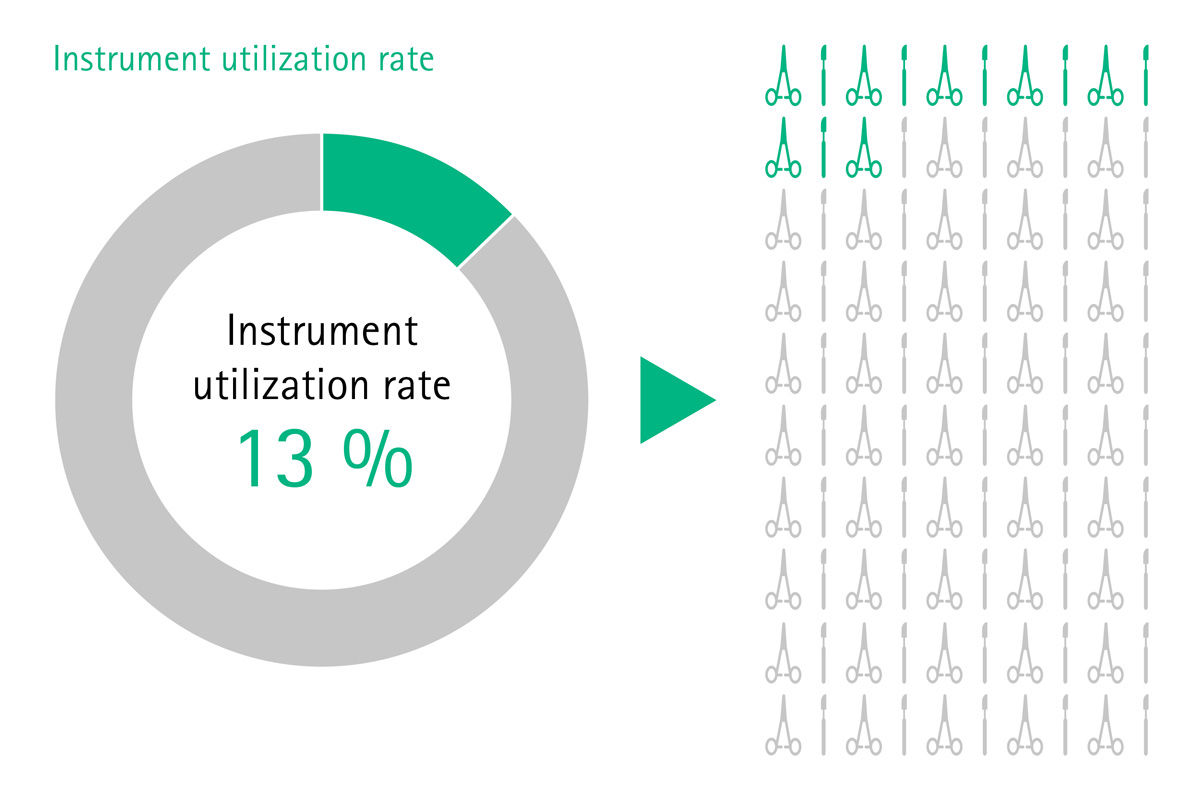
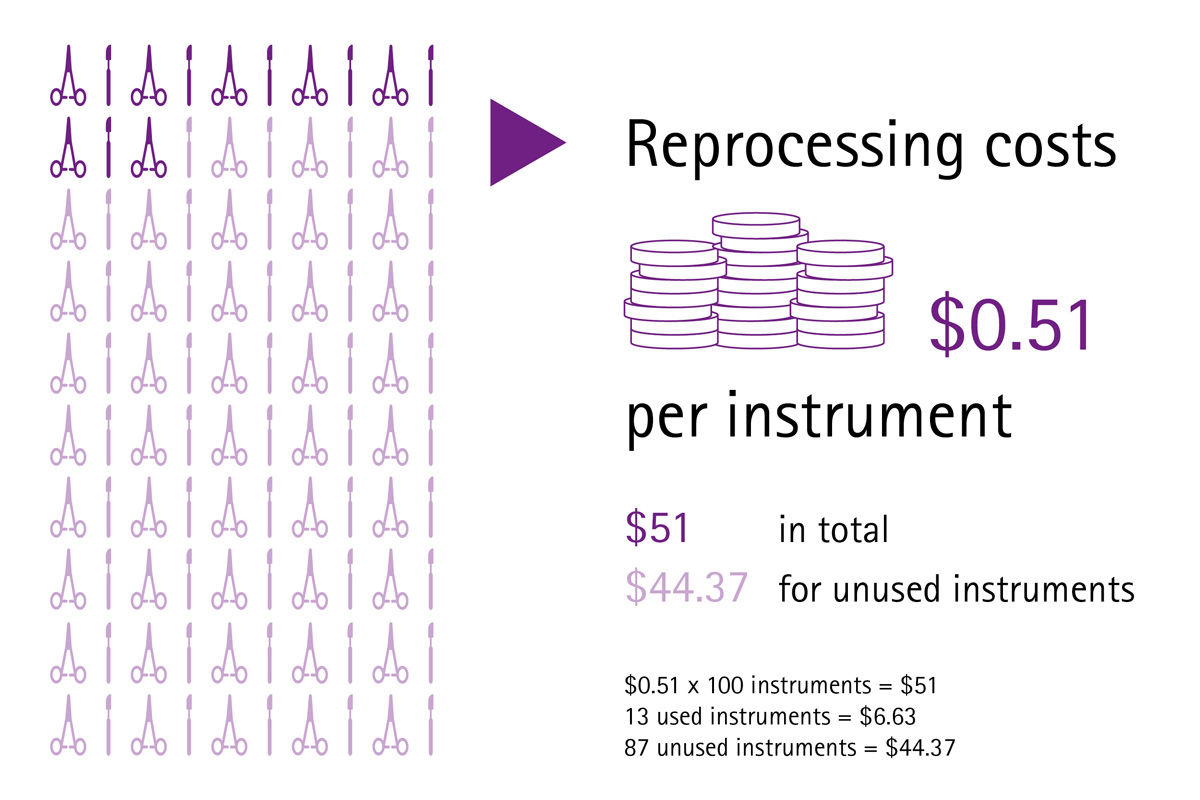
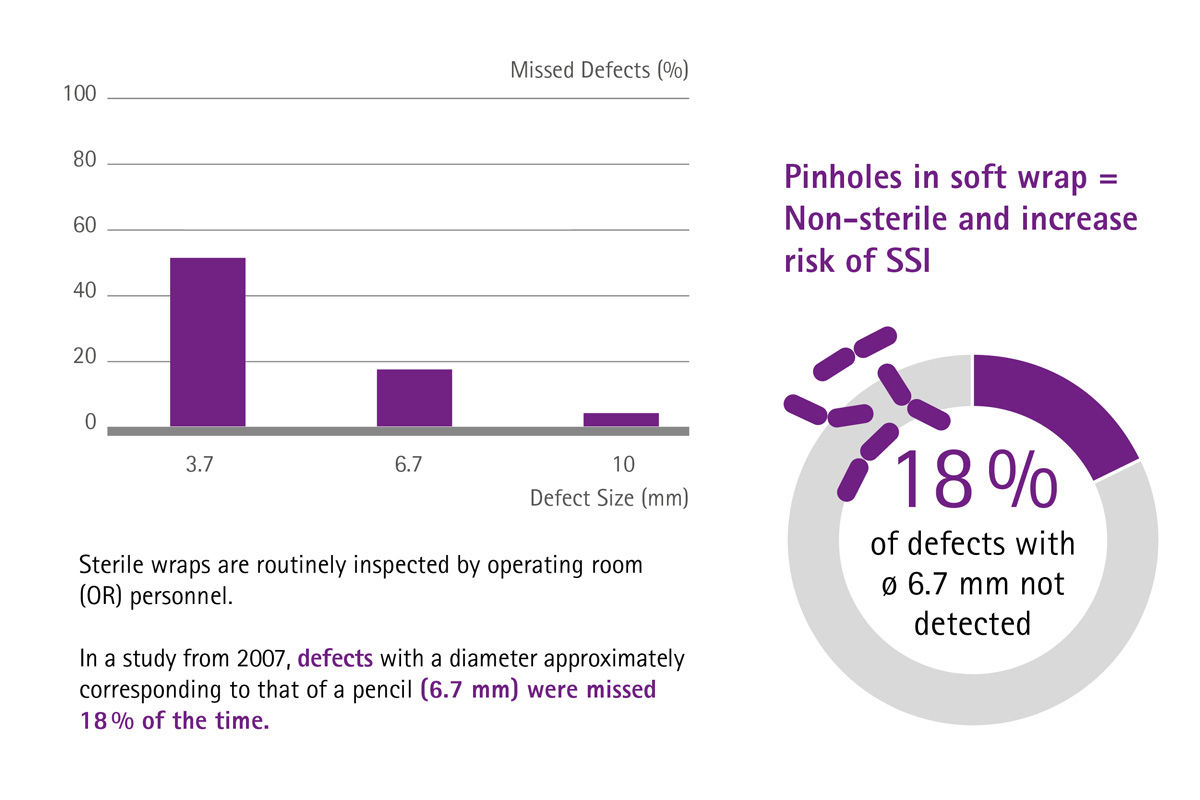
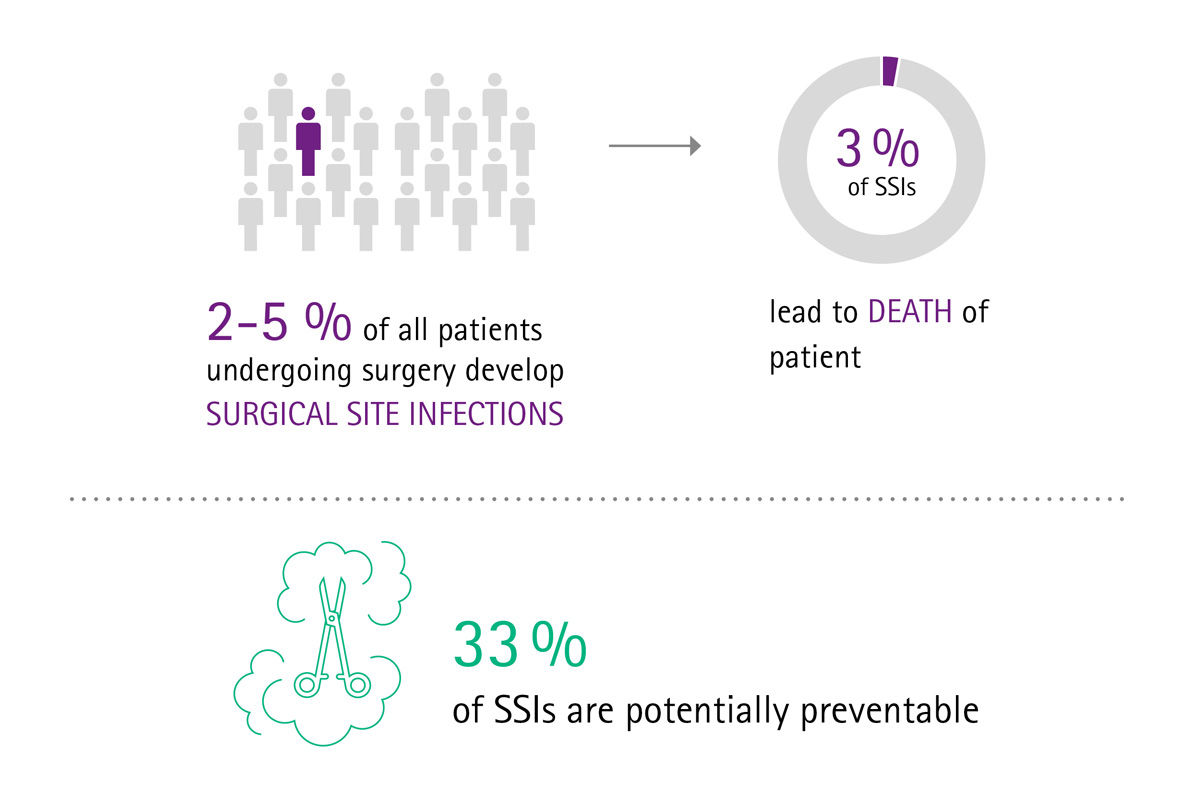
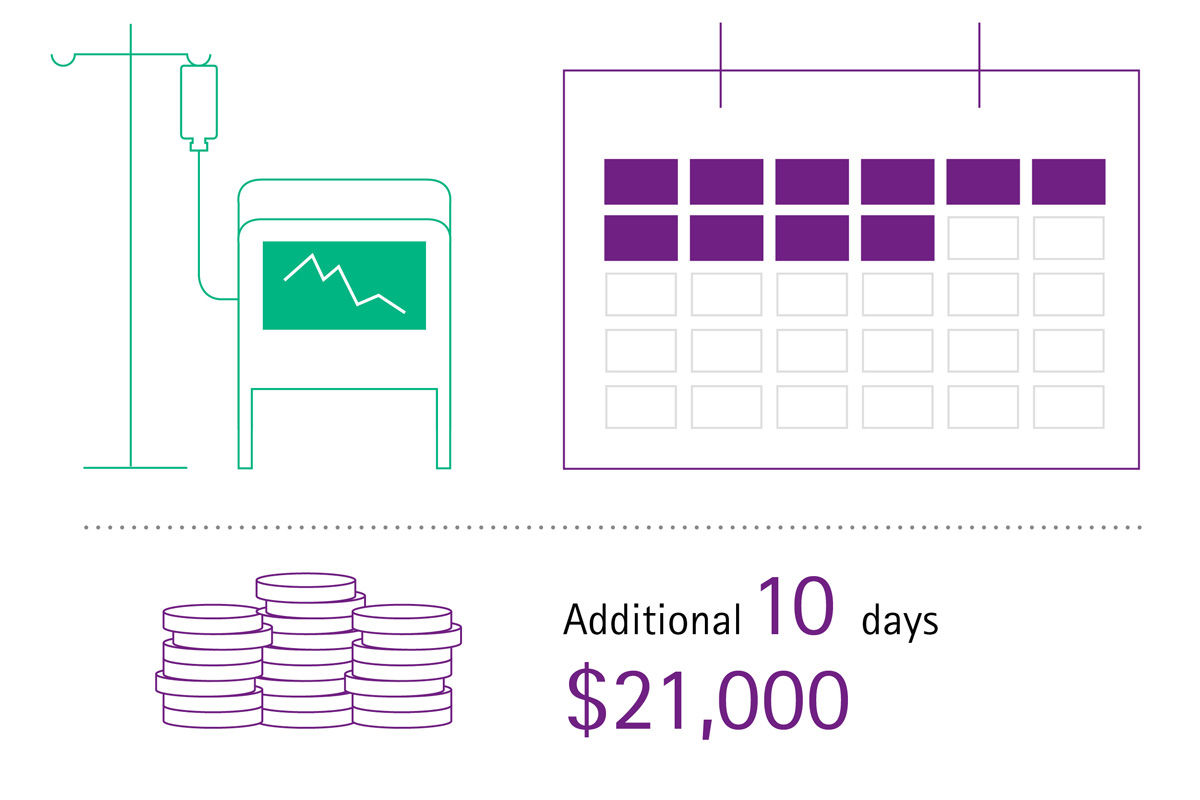
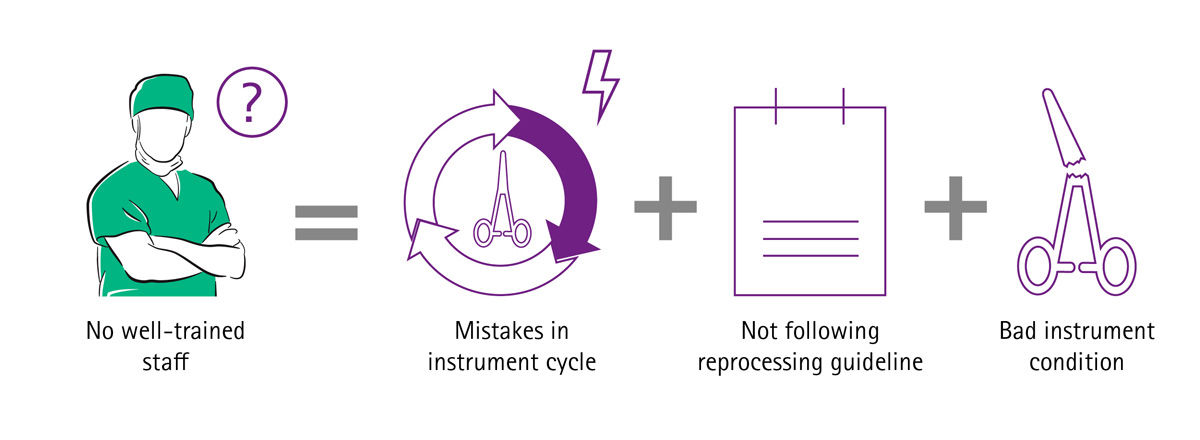

Imagine if your sterile goods management ran smoothly, surgical assets were in good condition, surgical sets were loaded with the correct number of instruments and reprocessed according to high quality standards, and therefore suitable for use in the OR and safe for treating patients. Imagine if you had cost transparency about the whole reprocessing cycle of the most valuable mobile asset in your hospital – the surgical instruments. There would be no complaints or extra costs for delays and interrupted surgeries, no frustration or stressed surgeons and staff, and there would be no prolonged hospitalizations because of SSIs either.
0%
of all surgical instruments are not suitable for use in the OR


Your feedback matters! Participate in our customer survey to help us enhance our website, products and services. Thank you for your support!